BioLaurus is Your Solution for Molecular Imaging
BioLaurus is a pre-clinical integrated molecular imaging research services provider to the biopharma industry. We’re working with leading biotech and pharmaceutical companies to accelerate their drug/device discovery and development stage research.
From consulting on study design through project execution, BioLaurus is focused on solutions that accelerate your research. Whether in vivo imaging on rodents, larger animals, or non-human primates we are there with you from start to finish. Our collaborative approach leverages your expertise and ours to create the optimum study. We use a clinical CRO business model that leverages key partnerships to maximize resources and minimize costs.
We operate in the major preclinical R&D segments of discovery stage efficacy, and bio-distribution studies and in development stage Safety Pharmacology PK, MOA, and Toxicology, and more. Much of our work is with clinical stage drugs where preclinical support studies are high stakes and timely results critical.
The greatest asset of BioLaurus is its integrative, solutions based approach and detailed operational plans to create, design and implement optimum studies in the most cost effective manner. We integrate imaging modalities including PE/CT, SPECT/CT, CT, micro-CT, ultrasound and optical (FLI and BLI) and combine with advanced computational imaging and analytics for drug quantification and PK, and anatomy. Therapeutic and device areas include oncology, cardiovascular, CNS Receptor Occupancy, advanced multi compartmental Drug PK, and in vivo cell PK.
Capabilities include contract cyclotron and radiochemistry expertise to do C-11 and F-18 radiotracers as well as Lu-177, Zr-89, Y-90, and other tracers. To learn more about BioLaurus capabilities explore our web site or walk through our online capabilities presentation.
Study Models
BioLaurus capabilities include a broad array of study models for both drug discovery and drug development stage therapeutics.
- CNS Receptor Occupancy/PK
- Opioid MOR, KOR
- Serotonin receptors
- GABA receptors, DAT
- Blood Brain Barrier permeability
- Cell tracking
- PLT survival and sequestration PK (rodent, rabbit, NHP)
- T-cell targeting in vivo (rodent)
- NASH (murine)
- Cardiomyopathy (rodent)
- Oncology, experience with over a hundred human tumor cell lines in a wide range of xenograft models in Nu/Nu, SCID, and NOD SCID strains. Orthotopic models include brain, breast, kidney, lung.
- Functional Imaging
- Perfusion
- Cardiac function
- Hepatobiliary function imaging
- Kidney function (renography)
- GI (enterocyte) function
- Angiography
- Reporter Gene models
- Luciferase/TK/ GFP
- Cell in vitro gene transfection/transduction
- In vivo (HDTV) gene transfection/transduction
- Bone Morphometry
- CT spinal fusion materials biodistribution, time course
- Micro-CT bone morphometry – bone destruction/regeneration
- Micro-CT bone morphometry – bone destruction/regeneration
Services We Provide
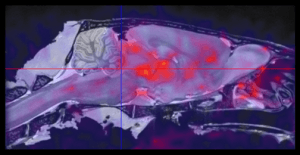
Molecular Imaging Services
Read More
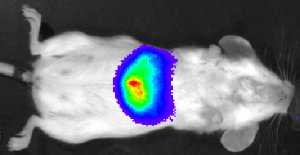
Imaging Modalities
Read More

Support Facilities & Equipment
Read More
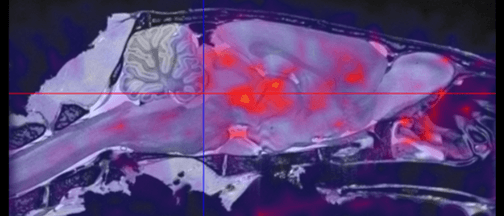
![Bone Spinal Fusion (fusion “putty” [green] replaced by new bone)](https://biolaurus.com/wp-content/uploads/2019/08/Spinal-Fusion-3-628x269.png)