Study Models
BioLaurus capabilities include a broad array of study models for both drug discovery and drug development stage therapeutics.
- CNS Receptor Occupancy/PK
- Opioid MOR, KOR
- Serotonin receptors
- GABA receptors, DAT
- Blood Brain Barrier permeability
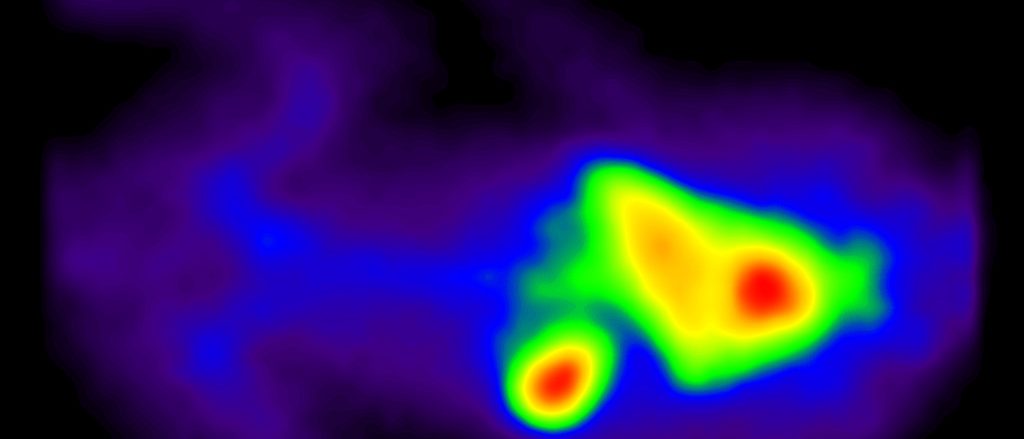
- Cell tracking
- PLT survival and sequestration PK (rodent, rabbit, NHP)
- T-cell targeting in vivo (rodent)
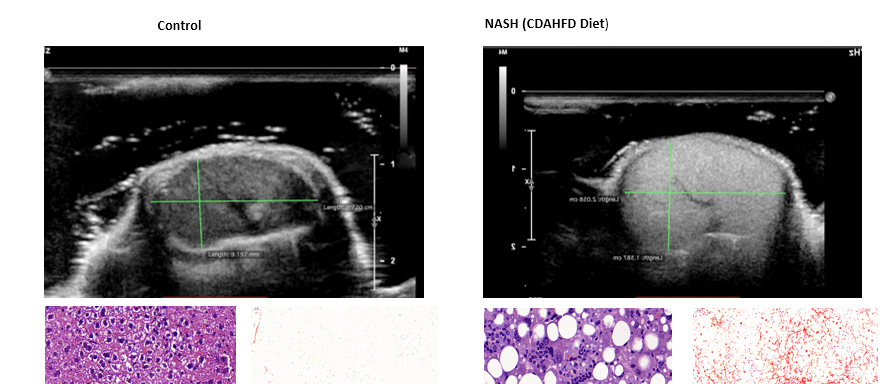
- NASH (murine)
- Cardiomyopathy (rodent)
- Oncology, experience with over a hundred human tumor cell lines in a wide range of xenograft models in Nu/Nu, SCID, and NOD SCID strains. Orthotopic models include brain, breast, kidney, lung.
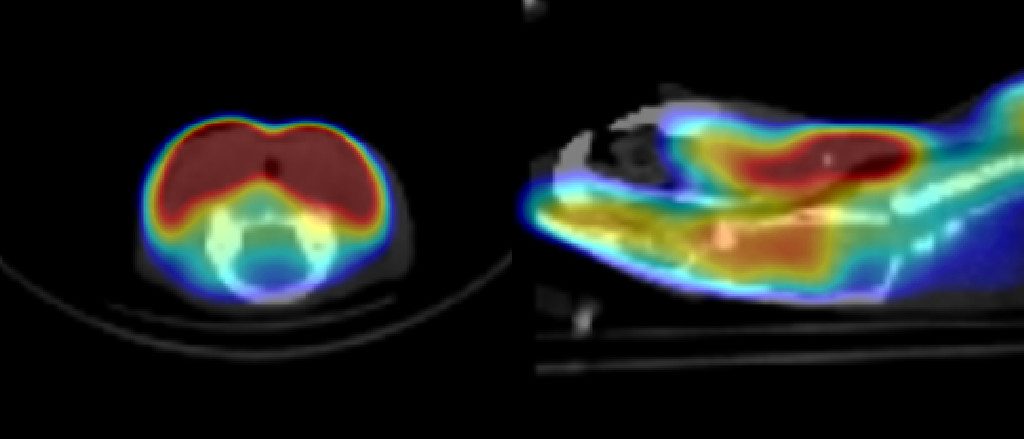
- Functional Imaging
- Perfusion
- Cardiac function
- Hepatobiliary function imaging
- Kidney function (renography)
- GI (enterocyte) function
- Angiography
- Reporter Gene models
- Luciferase/TK/ GFP
- Cell in vitro gene transfection/transduction
- In vivo (HDTV) gene transfection/transduction
- Bone Morphometry
- CT spinal fusion materials biodistribution, time course
- Micro-CT bone morphometry – bone destruction/regeneration