BioLaurus- A Molecular Imaging CRO
MOLECULAR IMAGING SOLUTIONS
BioLaurus is a pre-clinical integrated molecular imaging research services provider to the biotech/pharma Industry. We’re working with leading biotech and pharmaceutical companies to accelerate their drug/device discovery and development stage research.
From consulting on study design through project execution, BioLaurus is focused on solutions that accelerate your research. Whether in vivo imaging on rodents, larger animals, or non-human primates we are there with you from start to finish. Our collaborative approach leverages your expertise and ours to create the optimum study. We use a CRO Network business model that leverages key partnerships to maximize resources and minimize costs. We work with BioLaurus approved and AAALAC accredited and USDA registered vivarium along with and a team of in-house experts and consultants.
We conduct research in the major preclinical R&D segments of drug discovery and drug development including (but not limited to):
- Efficacy
- Bio-distribution studies
- Safety pharmacology PK, MOA, and Toxicology,
- Assay development
Much of our work is with clinical stage drugs where preclinical support studies are high stakes and timely results are critical.
We integrate imaging modalities including:
- PE/CT
- SPECT/CT
- CT
- Micro-CT
- MRI
- Ultrasound
- Optical (FLI and BLI)
with advanced computational imaging and analytics for drug quantification and PK, and bio-distribution.
Therapeutic and device areas include oncology, cardiovascular, CNS Receptor Occupancy, advanced multi compartmental Drug PK, and in vivo cell PK.
Capabilities include contract cyclotron and radiochemistry expertise to do C-11 and F-18 radiotracers as well as Lu-177, Zr-89, Y-90, and other tracers. To learn more about BioLaurus capabilities explore our web site or walk through our Capabilities slide deck.
Client Testimonials
I have worked with Mario on several programs. He is a gifted leader, very bright and a strong proponent of teamwork. Mario inspires people to outperform themselves and I have always been impressed with his professional demeanor, intelligence, and integrity.

Dr. Bourdon is a distinguished Bio-imaging expert with extensive experience and knowledge of imaging technologies. With great attention to detail, the study was only performed once, with all goals met – allowing for swift publication.

BioLaurus’ performance was outstanding in the study design and they provided trustworthy and insightful data and creative analysis techniques to get the most information from available data. I look forward to working with them in the future.
Services We Provide
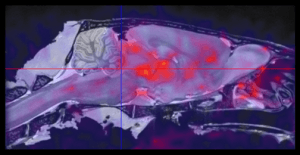
Molecular Imaging Services
Read More
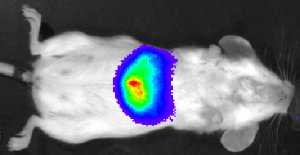
Imaging Modalities
Read More

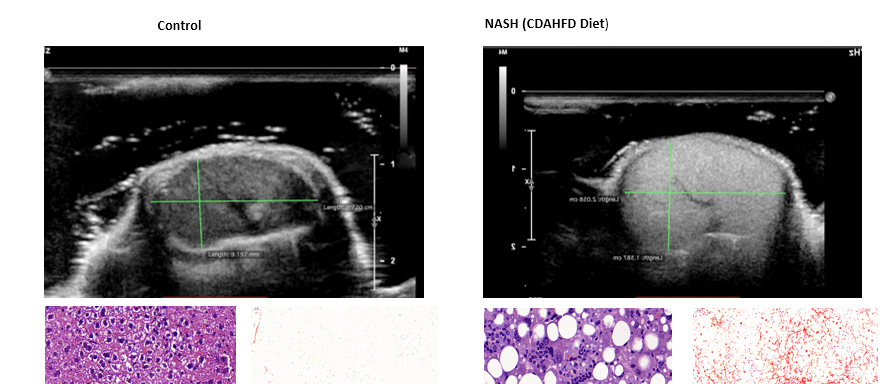
Support Facilities & Equipment
Read More
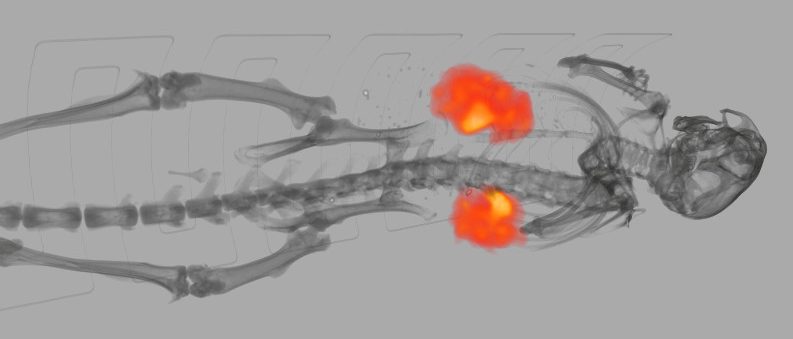
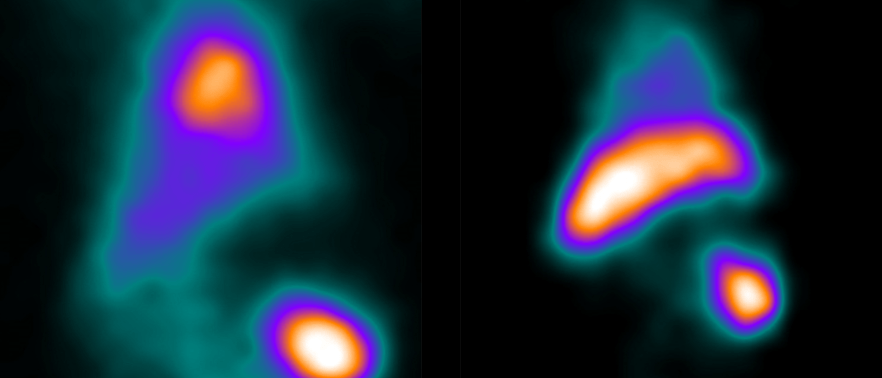
![Bone Spinal Fusion (fusion “putty” [green] replaced by new bone)](https://biolaurus.com/wp-content/uploads/2019/08/Spinal-Fusion-3-628x269.png)
