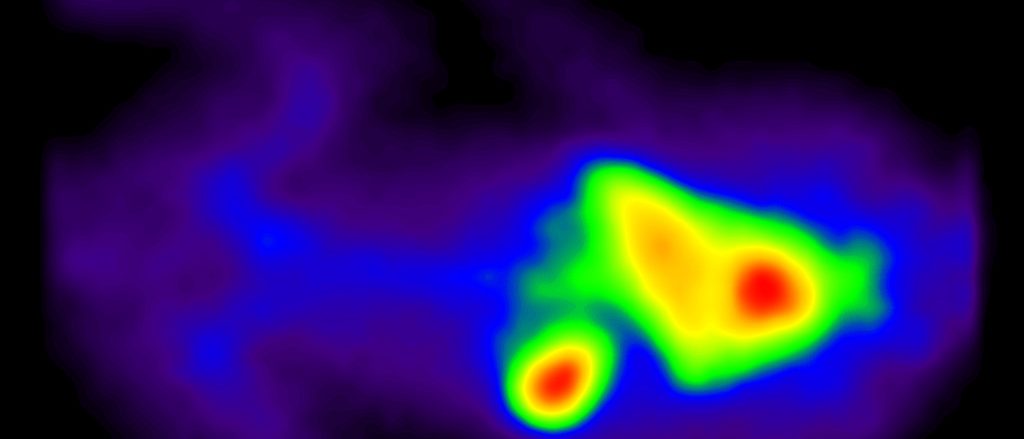
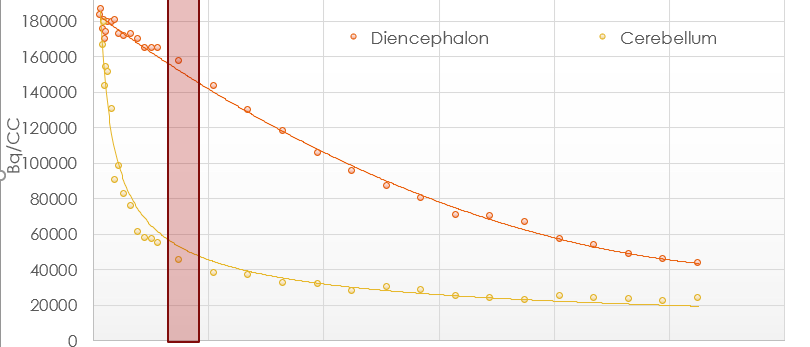
Imaging based assay development is a specialty of BioLaurus. We have developed assays for molecules ranging from small molecule drugs to antisence oligonucleotides, and biologics, to platelets and cells. These assays have ranged from CNS PET/CT opioid receptor occupancy assays with C-11 labeled opioid to SPECT platelet survival/sequestration PK with In-111 labeled platelet.
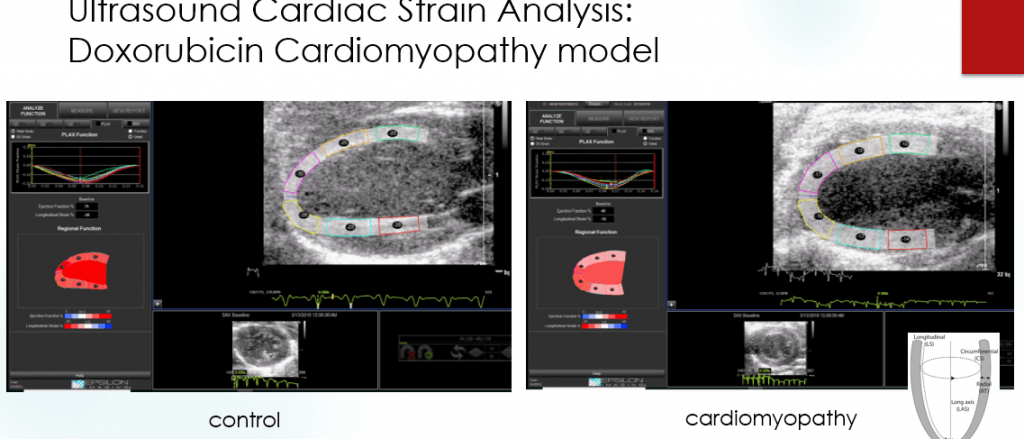
Assays include development of a rat cardiomyopathy model for early stage disease, an ultrasound assay of NASH in mice, ultrasound microbubble multiplexing for murine human tumor xenograft models, and a rat enterocyte CT functional imaging. We have the ability to do advanced C-11 and F18 chemistries as well as bi-functional DTPA, DOTA, PCTA chelators for In-111, Lu177, Y-90, and Zr-89 as well as expertise in fluorescence labeling and reporter gene assays. This expertise is integrated with over 100 years combined experience not only with assay development but the research background to fully understand the biologic context.