A wide range of drugs from heparin, to small molecules, antisence-oligonucleotides (ASA), peptides, and biologics have been found to reduce platelet (PLT) number with a variety of consequences including life threating thrombocytopenia.
Aster, R.H., Curtis, B.R., McFarland, J.G., Bougie, D.W., 2009. Drug-induced immune thrombocytopenia: pathogenesis, diagnosis, and management. Journal of Thrombosis and Hemostasis 7, 911–918.
Narayanan, P., Shen, L., Curtis, B.R., Bourdon, M.A., Nolan, J.P., Gupta, S., Hoffmaster, C., Zhou, F., Christian, B., Schaubhut, J.L., Greenlee, S., Burel, S.A., Witztum, J.L., Engelhardt, J.A., Henry, S.P., 2018. Investigation into the Mechanism(s) That Leads to Platelet Decreases in Cynomolgus Monkeys During Administration of ISIS 104838, a 2ʹ-MOE-Modified Antisense Oligonucleotide. Toxicological Sciences 164, 613–626.
Visentin,G.P. and Liu, C.Y., Drug Induced Thrombocytopenia Hematol Oncol Clin North Am. 2007 August ; 21(4): 685–vi
The major mechanism of drug-induced thrombocytopenia involves an immune process (DIITP). To make this determination is often difficult. Direct measures of anti-PLT and drug-PF4 antibodies must include both determination of IgM and IgG and results are rarely definitive. This is complicated by the generally low levels of antibodies that may be circulating, and timing between drug delivery and plasma sampling. Establishing this or any MOA requires critically imaging the sequestration process in vivo.
For decades, Thrombocytopenia has been measured experimentally and in the clinic by enumerating the concentration (k PLT/µl) of circulating PLT. There are three limitations in this method; first the PLT pool is mixed because new PLT are constantly being added, PLT have a life span in circulation, and old PLT are removed from circulation. Second, add to this complex normal kinetics the disease process and/or DIITP and third the inherent inability of measuring sequestration, the “were the circulating PLT go to”, by determining circulating PLT levels.
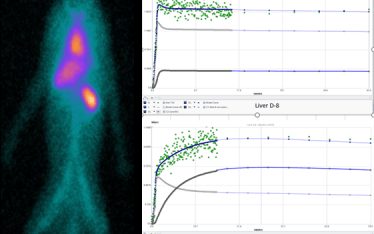
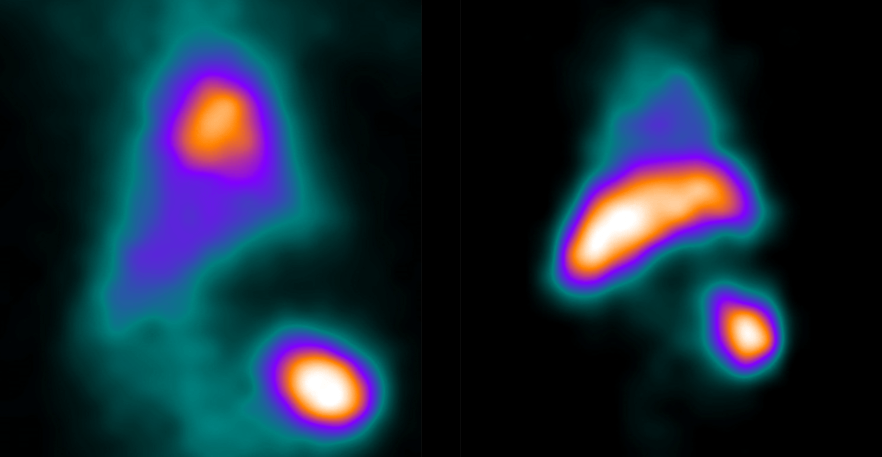
To determine the levels of circulating PLT and the levels of sequestration and as important where they sequester McAfee and Thakur pioneered the SPECT/gamma camera In-111 PLT tracking methods used today.
McAfee, J.G., Thakur, M.L., 1976. Survey of Radioactive Agents for in Vitro Labeling of Phagocytic Leukocytes. I. Soluble Agents. J Nucl Med 17, 480–487.
Wistow, B.W., Grossman, Z.D., McAfee, J.G., Subramanian, G., Henderson, R.W., Roskopf, M.L., 1978. Labeling of Platelets with Oxine Complexes of Tc-99m and In-111. Part 1. In Vitro Studies and Survival in the Rabbit. J Nucl Med 19, 483–487.
Tanaka, K.A., Katori, N., Kelly, A.B., Kotze, H., Marzec, U., Hanson, S.R., 2005. In vivo platelet redistribution and acute transient thrombocytopenia after eptifibatide injection in baboons. Thrombosis Research 115, 79–87
Narayanan, P., Shen, L., Curtis, B.R., Bourdon, M.A., Nolan, J.P., Gupta, S., Hoffmaster, C., Zhou, F., Christian, B., Schaubhut, J.L., Greenlee, S., Burel, S.A., Witztum, J.L., Engelhardt, J.A., Henry, S.P., 2018. Investigation into the Mechanism(s) That Leads to Platelet Decreases in Cynomolgus Monkeys During Administration of ISIS 104838, a 2ʹ-MOE-Modified Antisense Oligonucleotide. Toxicological Sciences 164, 613–626
SPECT imaging allows for the in vivo tracking of labeled PLT for up to 2 weeks however it is typically only necessary to track sequestration events over a 24 hour time course. Typically we do a 60 minute dynamic image scan beginning with PLT injection followed by scans at one or more later time points up to 24 hours. Dynamic imaging has opened up new opportunities to measure in real time and in vivo not only PLT concentrations in circulation and in any organ/tissue in the body but kinetic analysis.
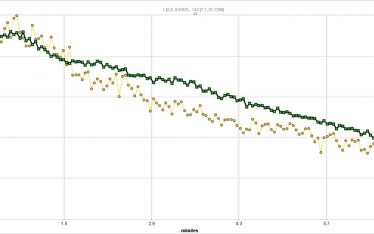
The standard means of comparing SUV (standard uptake value) and SUVR (standard uptake value ratio to reference tissue) is useful in making broad comparisons of sequestration between organs and between test subjects but cannot distinguish between circulating (free) and sequestered (bound) PLT in a target organ. In addition SUV and SUVR cannot directly provide information of kinetic rates of sequestration or bound PLT (PLT-sequestration “receptor” concentration) which might suggest a model of kinetics in each organ/tissue examined. That is the PK in one organ may be distinct from another organ with regard to mechanism. Indeed SUVR time activity curves for spleen and liver suggest this.
Currently, we are validating a PLT kinetic analysis method that is providing powerful insights into the different kinetics between spleen and liver and how different drugs can effect individuals differently in a population.