BioLaurus has developed multiple PET PK assays in rats. We are experts in the development of CNS PK assays and determination of the in vivo PK profile of opioid and their derivatives. The first opioid receptor assay developed targeted mu-opioid receptors (MOR) with the ligand C-11 carfentanil (CFN). With this assay PK data can be determined from time activity curves (TAC). In addition a high throughput in vivo ligand competition assay was developed. The MOR assay has been extended to other ligands including oxycodone and oxycodone derivatives in which these ligands are C-11 labeled. Currently we are developing PET KOR (kappa opioid receptor) assay to enable the examination of PK in MOR/KOR binding opioids.
The types of studies that can be performed can include cold ligand multidose studies with ligand added prior to radioligand or co-injected. The choice of pre-dosing or co-injection depends in part on the goals of the study. If durability of receptor occupancy or sensitivity to low level receptor occupancy by a low affinity or permeability competitive ligand is a focus then pre-dosing likely is preferred. Where the affinity of the cold ligand: hot ligand, are within 100 to 1000-fold of each other direct co-injection competition can be done.
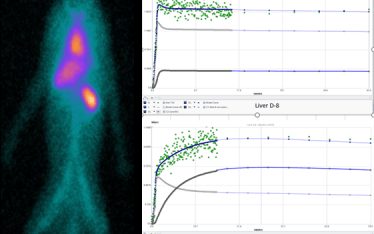
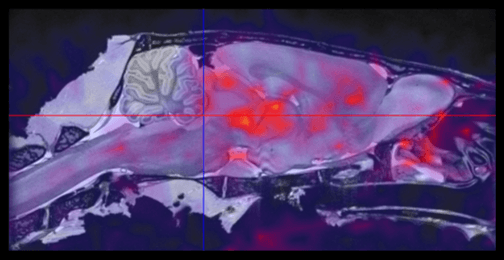
The TAC data for PK analysis is derived from PET dynamic images of high specific activity C-11 CFN injected (nano-mol mass) into normal rats. The TAC are generated over a 60 to 90 minute time course. In the example shown here two TAC are shown, one for the C-11 CFN alone and second C-11 CFN TAC in rats pre-dosed with the inverse agonist naloxone. The naloxone completely blocks C-11 CFN binding in the brain. The PK data is generated in PKIN by analyzing TAC for high receptor MOR regions (diencephalon), the reference region ( cerebellum) and the arterial input function. While this discussion has focused on ligand receptor occupancy this same PET assay can be used to do displacement studies. Displacement studies are powerful tool in dissecting multi-receptor interactions for example whether more than one MOR receptor is involved. In these studies cold ligand is added in high concentration following C-11 CFN tracer.
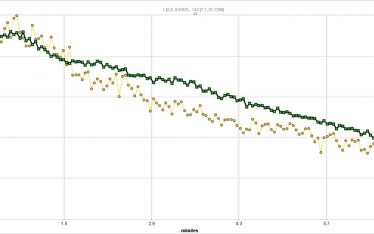
A limitation of the TAC PET assay is the short half-life of C-11 (20 minutes) which means only one TAC can be generated per synthesis. To allow for higher throughput a “window” assay was devised that was derived from dynamic TAC data. In the window assay a short time window is selected for data acquisition at a point just beyond the maximum ROI:cerebellum SUVR TAC point. As long as the window is fixed, and the C-11 tracer concentration (nanomole/kg) is fixed the SUVR can reliably by compared for up to 4 injections/synthesis. By comparing inhibitor concentration curves a standard IC30 can be determined and the cold ligand IC30 concentration compared. While very useful in rapidly comparing compounds binding the target receptor this approach does not generate PK data.