Therapeutic drugs targeting a wide range of central nervous system (CNS) disorders work by interacting with CNS receptors. Developing those drugs inevitably leads to needing to understand Drug/ Receptor PK/PD and comparison of receptor occupancy (RO) of drug candidates. PET imaging and receptor occupancy (RO) analysis can measure receptor-drug kinetics and compare the in vivo activities of drug candidates but there are pitfalls. Here we will share a few insights that help toward a successful study.
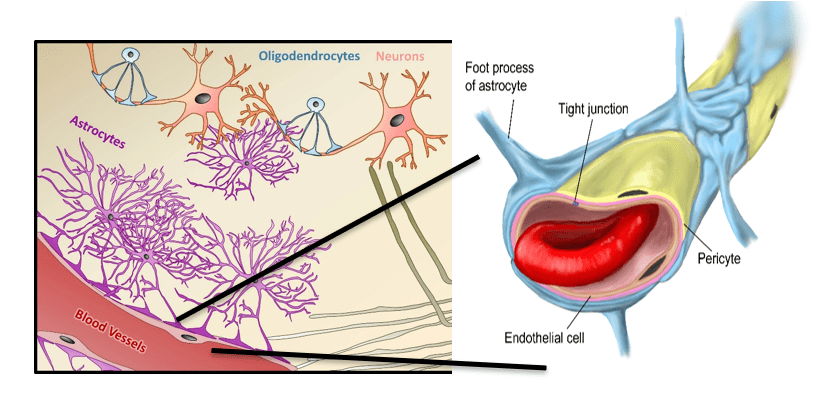
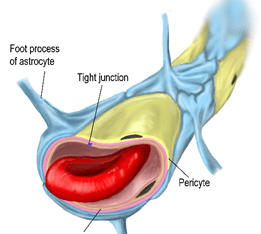
Step 1. Always keep in mind that the brain is a unique environment with unique tissue compartments. The blood brain barrier (BBB) compartment has enormous impact on PET RO assays and interpretation of results. This will be dealt with in detail in a separate part of the series. Unlike any other vascular feature in the body, the BBB is highly selective in allowing entry of molecules into the brain. Only small molecules that are lipid soluble such as caffeine, or those such as glucose with transporters (GLUT-1) can enter the brain. Even drugs that based on lipid solubility should enter the brain can be excluded partially or completely via efflux transporters like P-gp. While lipid solubility and BBB transporters are little changed between species efflux transporters are another story.
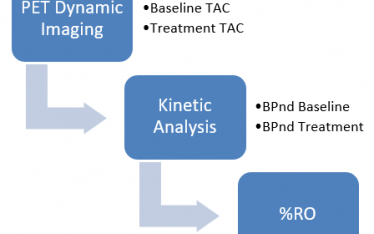
Step 2. Define the problem carefully. What really needs to be measured and how? Usually ligand-receptor binding kinetics can be modeled as a plasma compartment (CP) and two tissue compartments, BBB (C1), and parenchymal receptor compartment (C2).
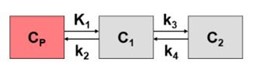
Since RO is typically described as
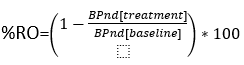
the BPnd or k3/k4 must be determined from radiotracer kinetics. The radiotracer PET assay has two components, 1. the radiotracer – receptor binding component, what is measured, and 2. the challenge or comparator drug conditions. This component considers drug comparator concentration(s), dose route, time course, and sequence (predosing, co-injection, post dosing of comparator and radiotracer).
Another critical question is what type of assay answers your question? What do I mean? The PET assay can be used to directly examine the radiotracer receptor kinetics or extended to compare drugs binding to the receptor of interest.
Indirect receptor binding kinetics RO. The radiotracer ligand – receptor binding is the sole focus. You may want to know if other drugs are competitors or inhibitors of the radiotracer ligand – receptor binding and you may also want to examine the effects of a drug that does not directly compete for the receptor but effects Cp or BBB.
The comparator RO assay compares receptor occupancy of the radiotracer ligand to one or more other unlabeled drugs. The goal is to set up reproducible in vivo conditions that then allow comparison of the readout, RO, between radiotracer and comparator drug and between comparator drugs. Remember we are not directly measuring the comparator drug kinetics, only its effects on radiotracer RO. It is often assumed that the comparator drug does not affect the radiotracer kinetics but there are situations where this is not true Getting this concept wrong is the greatest source of failure. Seek advice from those with experience.
Step 3. For your receptor target is there an “off the shelf” assay or does a new assay need to be developed? A quick literature search should suffice to get some background. Typically, at BioLaurus we will draw on our previous experience and supplement that with a targeted literature search. Whether off the shelf or not there are a series of assay evaluations including controls that must be considered. There are no short cuts. Design the study for its purpose, proof of concept, screening, or detailed PK.
Step 4. Select the drug that will be used as the radiotracer in measuring the receptor binding. Often this is aided by the available literature and your PET consultant or CRO should be able to give you options. Here are a few criteria for selecting a radiotracer.
- Is the radiotracer specific for the receptor target? What positive and negative controls are there?
- Can the candidate radiotracer be radiolabeled to high specific activity?
- Can the radiotracer pass the BBB and does enough cross to measure specific receptor binding?
Step 5. Study Design
We will go into more depth in other parts of this series but here we will touch on a few key considerations.
- Choice of test subject. As most drugs being tested are intended to treat humans the animal model must mimic the human as closely as possible. This means any significant species difference in BBB or receptor must be known and either mitigated or require the choice of another model.
- The ligand/receptor profile is the easier to deal with. In vitro and genomic analysis usually provides the key information on cross species specificity, Kd, and homology. Be aware that PK data from in vitro studies or blood PK data often do not reflect in vivo receptor kinetics.
- The BBB is often more problematic. Lipid solubility and efflux transporters are the biggest challenges. If the radiotracer does not have a transporter, or is not lipid soluble, or is an efflux transporter ligand too little radiotracer enters the brain to reliably measure RO.
To summarize, the path to a successful PET RO Assay begins with good planning up front. Like all assays, it is important to work with people who have previous experience with doing PET assays.